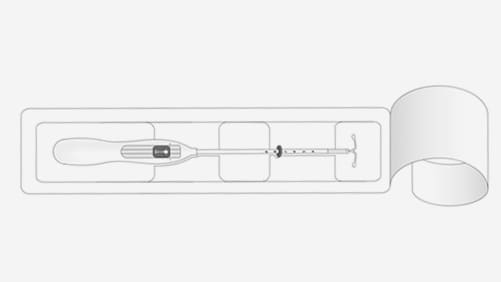
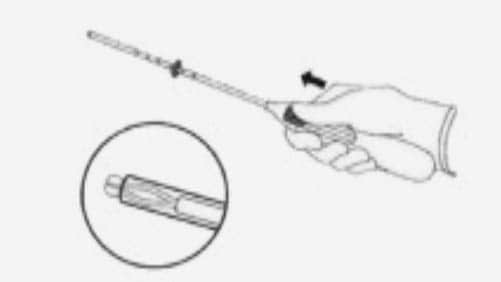
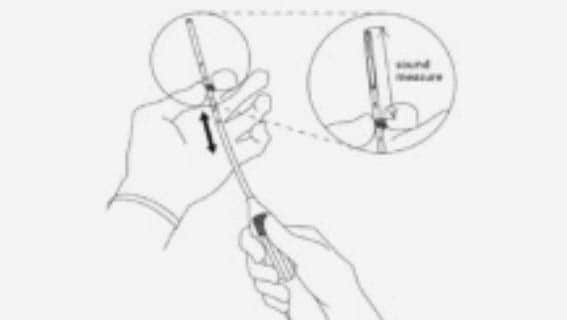
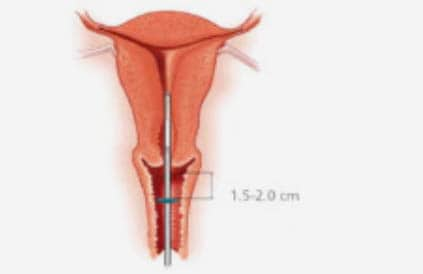
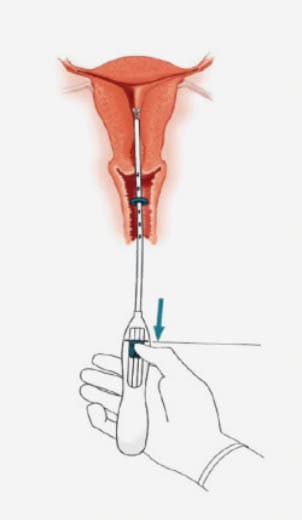
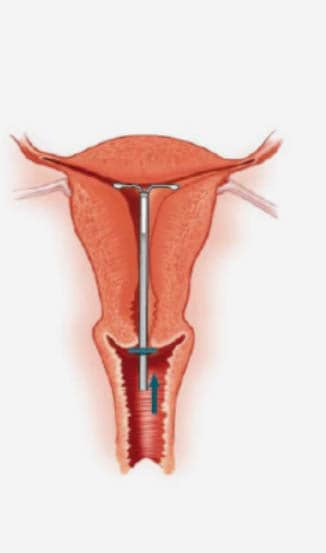
Step-by-step instructions to guide you through the preparation and insertion of an IUS.
The summary of product characteristics should be referred to before inserting an IUS.
Key considerations when preparing for an IUS insertion:1
- Obtain informed consent and arrange for chaperone.1
- An appropriately trained assistant should be present (to monitor & support the patient and assist in an emergency).2
- Basic risk assessment includes gathering information about previous intrauterine procedures. Patients who have had previous adverse events during insertion are more likely to have them again.3
- Pulse rate and blood pressure should be documented.3
- The need for pain relief during insertion should be discussed with the patient.3
- Routine antibiotic prophylaxis is not recommended pre-insertion. However, for women at increased risk of STIs, in whom testing has not been completed, prophylactic antibiotics are advised.1
- Johnson BA. Insertion and removal of intrauterine devices, American Family Physician. January (2005). Return to content
- FSRH. Intrauterine Contraception [online] October 2015. Available from: https://www.fsrh.org/standards-and-guidance/documents/ceuguidanceintrauterinecontraception/. Return to content
- Mirena® Summary of Product Characteristics. Return to content
Inserting an IUS – the procedure1
- For women with symptomatic pelvic infection, postpone insertion and treat infection. Treatment should be complete before another attempt at insertion.
- A pelvic examination should be performed prior to inserting the device, to assess the size, shape, position and mobility of the uterus.
- Assessment of uterine size by sound measure.
- Forceps (tenaculum) are used to stabilise the cervix during insertion and reduce risk of perforation.
- Some inserters offer para-cervical block for the procedure.
- Many inserters use local anaesthetic gel on the cervix.
- Documentation should be made in the case notes of pre- and post-insertion counselling, the procedure, the type of device inserted, and any adverse events.
- Johnson BA. Insertion and removal of intrauterine devices, American Family Physician. January (2005). Return to content
Practical aspects of inserting an IUS1
Potential problems at the time of insertion
Vasovagal collapse/cervical shock:
- Stop the procedure, lower the head and/or raise the legs. The IUS may need to be removed. An assistant should monitor observations. Ensure a clear airway. ‘ABC approach’ in basic resuscitation.
- Oxygen and suction as required. Consider use of atropine IV/IM for persistent bradycardia. Consider use of adrenalineIM 1:1000 for management of anaphylaxis.
- Automated external defibrillator (AED) should be available. Arrange ambulance transfer if there is no improvement.
Syncope:
- Syncope may be experienced at insertion secondary to vagal stimulation from the cervix.1 Bradycardia is more common in nulliparous women.2
Suspected uterine perforation:1
- If perforation is suspected at the time of insertion, stop the procedure and monitor observations. Ultrasound or plain abdominal film to locate the device should be arranged as soon as possible, depending on device.
- Mirena® Product Monograph Return to content
- Farmer M, Webb A; Intrauterine device insertion-related complications: can they be predicted? J Fam Plann Reprod Health Care. 2003 Oct;29(4):227-31. Return to content
Post-insertion advice
Provide patients with a patient information leaflet and advice post-insertion of IUS is an important component of patient care.
Key advice to be offered includes:1
Expect mild cramping. This usually resolves itself but if you have any concerns please contact your healthcare professional
Informing the patient on how to feel the threads (e.g. before the next episode of sexual intercourse and after her next menses) and advise her to seek medical advice if she is unable to feel them.
It may also be worthwhile discussing:
Bleeding
Infection
If the patient is feeling well enough to leave and is comfortable
Ensure documentation completed
A follow-up appointment with the patient for 6 weeks’ time
- Mirena® Product Monograph Return to content
Good practice points for documentation1
Detailed documentation is a key component of good practice in IUS insertion. Important points to document include:
- Faculty of Sexual and Reproductive Healthcare. Service Standards for Record Keeping in Sexual and Reproductive Healthcare Services, July 2019.
Available at: https://www.fsrh.org/standards-and-guidance/documents/fsrh-service-standards-for-record-keeping-july-2019/. Return to content
Mirena® (52 mg Levonorgestrel) insertion, removal and replacement
Step-by-step video guidance for the preparation, insertion, removal and replacement of the Mirena® IUS.
Recommendations
Reporting adverse events and quality complaints
If you want to report a side effect or quality complaint, please contact your health care professional (e.g. physician or pharmacist) or The Health Products Regulatory Authority, Reports can also be reported directly to Bayer through this link or by emailing directly on adr-reland@bayerhealthcare.com